Lithium batteries have become increasingly popular over the years as the demand for high-performance batteries continues to rise. These batteries are used to power a variety of electronic devices, including smartphones, laptops, and even electric vehicles.
One such lithium battery that has gained popularity is the 14500 Lithium battery. This high-capacity rechargeable battery is widely used in devices that require a lot of power. Its small size and high energy density make it ideal for portable electronics such as flashlights and toys.
The manufacturing process of these batteries, including the 14500 Lithium battery, is fascinating and involves several stages, from the extraction of lithium to the assembly of the battery cell. With advancements in technology, the manufacturing process for these batteries is becoming more efficient, resulting in more cost-effective and powerful batteries.
As the demand for lithium batteries continues to grow, the production of these batteries will undoubtedly play an essential role in powering the future.
Stage 1: Extraction of Lithium
The first step in lithium battery manufacturing is extracting ore from its natural sources. This metal is found in rock formations, brines, and clays. The most common source of lithium is brine, which is pumped from underground reservoirs. The brine is then evaporated using solar energy, and the remaining concentrate is treated to extract lithium.
Stage 2: Processing of Lithium
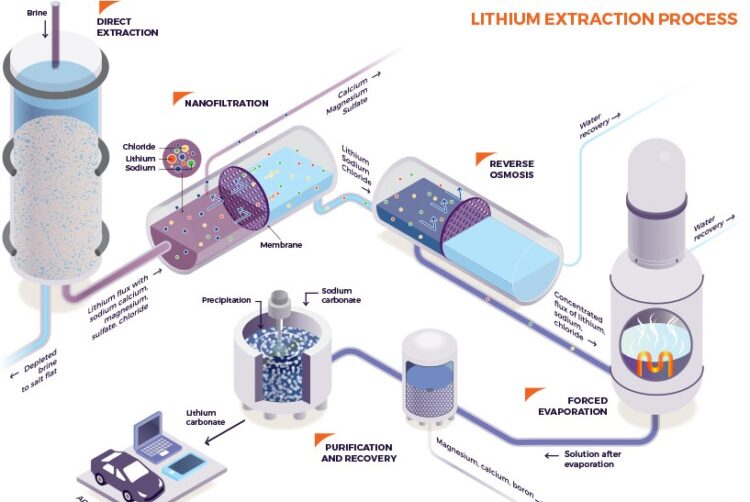
Once lithium is extracted, it undergoes several processing stages to produce a high-purity lithium compound. The processing stages include crushing, roasting, and leaching. Crushing involves grinding the extracted material into small particles. Roasting involves heating the crushed material to remove impurities, and leaching involves dissolving the roasted material in water.
Stage 3: Production of Cathode Materials
The next stage in battery manufacturing is the production of cathode materials. Cathode materials are the positive electrode in a battery, and they are typically made of lithium cobalt oxide, lithium iron phosphate, or lithium nickel manganese cobalt oxide. These materials are produced by mixing the appropriate metal oxides, carbon, and binders in a specific ratio and heating them to high temperatures.
Stage 4: Production of Anode Materials
Anode materials are the negative electrode in a lithium-ion battery, and they are typically made of graphite or silicon. The production of anode materials involves mixing the appropriate carbon and binders and then coating the mixture onto a copper foil substrate. The coated substrate is then dried and heated to high temperatures to form the anode material.
Stage 5: Assembly of the Battery

Once the cathode and anode materials are produced, they are assembled into a battery cell. The cell assembly involves placing the anode and cathode materials with a separator in between them and then rolling them together into a cylinder. The cylinder is then inserted into a metal casing, and the space between the cylinder and the casing is filled with an electrolyte solution.
Stage 6: Testing and Packaging
After the battery cell is assembled, it undergoes several testing stages to ensure that it meets the required specifications. The testing stages include measuring the voltage, capacity, and impedance of the cell. Once the cell passes the testing stages, it is packaged into a final product, such as a battery pack for an electric car or a battery for a smartphone.
Conclusion
The process of lithium battery manufacturing involves several stages, from the extraction of lithium to the assembly of the battery cell.
Each stage requires specialized equipment and knowledge, and the final product is a high-performance battery that powers our electronic devices. The demand for lithium batteries is increasing, and manufacturers are continually improving the manufacturing process to produce more efficient and cost-effective batteries.
 Hi Boox Popular Magazine 2024
Hi Boox Popular Magazine 2024



